FDA’s Rare Disease Endpoint Advancement (RDEA) Pilot Meeting Program is accepting RDEA proposals for this quarter until September 30, 2023. The RDEA Pilot Meeting Program is designed to support novel endpoint efficacy development for drugs that treat rare diseases by providing a mechanism for sponsors to collaborate with FDA throughout the efficacy endpoint development process. […]
Read MoreThe Food and Drug Administration’s (FDA) Office of Orphan Products Development (OOPD) is pleased to announce availability of funds for fiscal years (FY) 2023 – FY 2025 to support clinical trials for rare diseases and conditions. The purpose of this funding opportunity announcement is to fund clinical trials of products evaluating efficacy and/or safety in […]
Read MoreThe Rare Diseases University (RDU) is a physician-oriented, interactive, digital Medical Education program. It was designed to address the urgent need for better patient diagnosis and care for patients living with rare diseases around the world.The content is developed with the assistance of a faculty of doctors working in the field of rare diseases, and […]
Read MoreRare Diseases International (RDI) and the Permanent Mission of Spain to the United Nations in New York are organizing the hybrid event “Engaging the UN System and Member States to Achieve UHC for PLWRD: A Blueprint for Leaving No One Behind” on Thursday, 21 September, 2023, from 13:00 to 14:30 EST. The event is co-organized […]
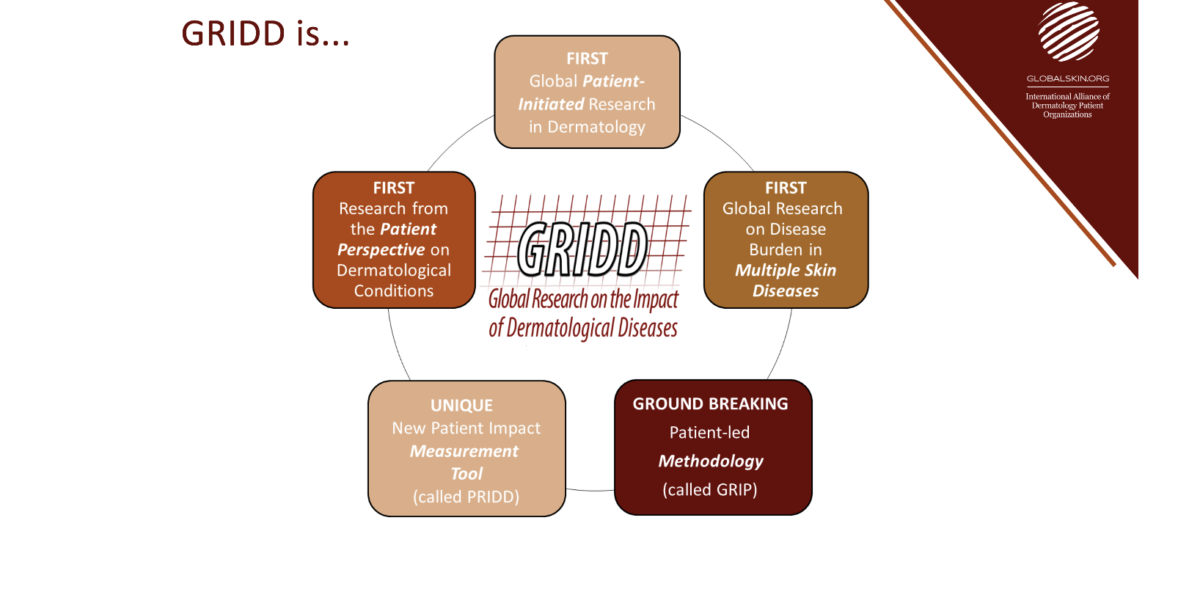
Read MoreThe Global Research on Impact of Dermatological Diseases (GRIDD) project is a global initiative focused on demonstrating the impact of skin conditions on patients’ lives. Its purpose is to gather worldwide data on dermatological diseases directly from those affected. The heart of GRIDD lies in the new Patient-Reported Impact of Dermatological Diseases (PRIDD) measure, a […]
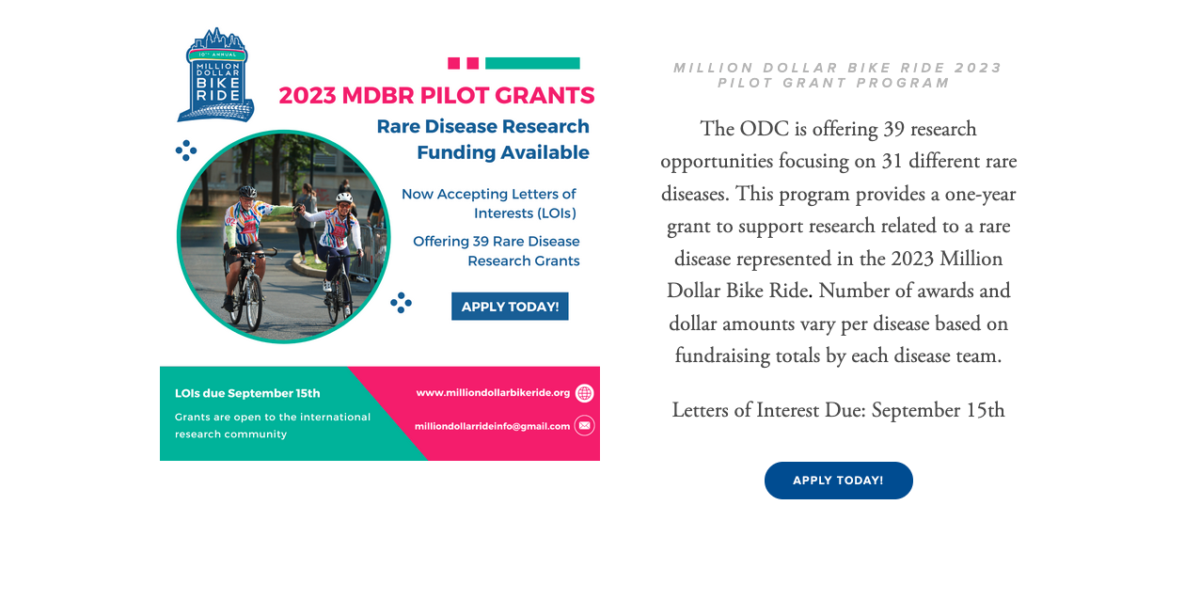
Read MoreThe Orphan Disease Center (ODC) is offering 39 research opportunities focusing on 31 different rare diseases. The program provides a one-year grant to support research related to a rare disease represented in the 2023 Million Dollar Bike Ride. Number of awards and dollar amounts vary per disease based on fundraising totals by each disease team. […]
Read More“Rare on air” is a series of monthly podcasts developed by EURORDIS showcasing different interviews with people who live with a rare diseases, advocates and experts on rare disease policy. The podcast explores the unique experiences, challenges and successes of people from the rare disease community, and investigates how we can build a better world […]
Read MoreIRDiRC is delighted to share the manuscript “How to START? Four pillars to optimally begin your orphan drug development“, developed by the members of the IRDiRC Task Force “Orphan Drug Development Guidebook (ODDG)” together with the members of the Therapies Scientific Committee (TSC). The paper proposes a checklist for orphan drugs named START – a […]
Read MoreDon’t miss out the European Haemophilia Consortium (EHC) Conference that will take place this year on 6-8 October, 2023, in Zagreb, Croatia, where IRDiRC Interdisciplinary Scientific Committee (ISC) Vice Chair, Marc Dooms (University Hospitals Leuven, Belgium), will present at the European Rare and Inhibitor Network (ERIN) session: “Basket trails – changes that we need”. Check […]
Read MoreRare disease research presents unique challenges due to the small size of the populations of people living with these conditions. Collecting and curating large study cohorts in rare diseases presents a real problem, which can limit understanding of the clinical spectrum of disease. The SANOFI Rare Disease Registries represent more than 30 years of real-world […]
Read More